nataliezzz
Senior Member (Voting Rights)
Objective versus subjective excessive daytime sleepiness in OSA: Quantifying the impact of fatigue
Morris S Gold, Riccardo A Stoohs, Avram R Gold
https://www.sciencedirect.com/science/article/abs/pii/S1389945726001061
Background
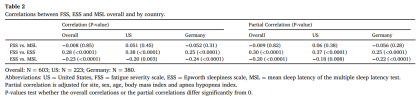
In patients with obstructive sleep apnea (OSA), excessive daytime sleepiness (EDS) is typically measured either objectively with the mean sleep latency (MSL) of the multiple sleep latency test (MSLT), or subjectively with the Epworth sleepiness scale (ESS). These measures correlate only mildly with each other and differ greatly in their correlations with comorbidities and outcomes associated with OSA. To improve our rudimentary understanding of the differences between objective and subjective EDS, we compared the quantitative impact of fatigue on objective and subjective EDS using the fatigue severity scale (FSS) to measure fatigue.
Methods
We identified 603 patients with OSA in a US site and a German site who had completed a MSLT, ESS and FSS between 2008 and 2023. The relationships of FSS with MSL and ESS were assessed with simple summary statistics, correlation and linear regression.
Results
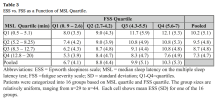
MSL and the FSS were uncorrelated, i.e. fatigue has no impact on objective EDS. This allowed us to simultaneously quantify distinct impacts of objective EDS (MSL) and fatigue (FSS) on subjective EDS (ESS). MSL and FSS were found to have separate and roughly equal impacts on ESS. Thus, whereas MSL is a pure measure of EDS, ESS measures a roughly equal mix of EDS and fatigue.
Conclusions
In patients with OSA, EDS should be measured with the MSLT. A fatigue scale should replace the ESS, which measures an uninterpretable mix of EDS and fatigue. The role of fatigue in the clinical profile of patients with OSA must be fully established.
Morris S Gold, Riccardo A Stoohs, Avram R Gold
https://www.sciencedirect.com/science/article/abs/pii/S1389945726001061
Background
In patients with obstructive sleep apnea (OSA), excessive daytime sleepiness (EDS) is typically measured either objectively with the mean sleep latency (MSL) of the multiple sleep latency test (MSLT), or subjectively with the Epworth sleepiness scale (ESS). These measures correlate only mildly with each other and differ greatly in their correlations with comorbidities and outcomes associated with OSA. To improve our rudimentary understanding of the differences between objective and subjective EDS, we compared the quantitative impact of fatigue on objective and subjective EDS using the fatigue severity scale (FSS) to measure fatigue.
Methods
We identified 603 patients with OSA in a US site and a German site who had completed a MSLT, ESS and FSS between 2008 and 2023. The relationships of FSS with MSL and ESS were assessed with simple summary statistics, correlation and linear regression.
Results
MSL and the FSS were uncorrelated, i.e. fatigue has no impact on objective EDS. This allowed us to simultaneously quantify distinct impacts of objective EDS (MSL) and fatigue (FSS) on subjective EDS (ESS). MSL and FSS were found to have separate and roughly equal impacts on ESS. Thus, whereas MSL is a pure measure of EDS, ESS measures a roughly equal mix of EDS and fatigue.
Conclusions
In patients with OSA, EDS should be measured with the MSLT. A fatigue scale should replace the ESS, which measures an uninterpretable mix of EDS and fatigue. The role of fatigue in the clinical profile of patients with OSA must be fully established.