Neuroendocrine signature of ME/CFS: Meta-analytic evidence for bioactive cortisol deficit and exaggerated feedback sensitivity
Abstract
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is a major clinical challenge as a complex multisystemic disorder with no well-established pathophysiological mechanism, characterized by persistent fatigue and post-exertional malaise, along with unrefreshing sleep, cognitive impairment, and impaired stress recovery.
Despite decades of investigation into the hypothalamic-pituitary-adrenal (HPA) axis, a definitive neuroendocrine hallmark has remained elusive due to inconsistent findings across various cortisol matrices. Therefore, this systematic review and meta-analysis aimed to provide an integrated understanding of HPA-axis regulation in ME/CFS.
We identified 46 case–control studies (comprising 46 independent datasets, including 12 pharmacological challenge studies), involving 1388 ME/CFS patients (71.9% female; mean age 37.3 ± 6.2 years) and 1349 matched healthy controls.
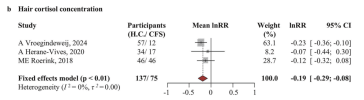
Meta-analyses showed lower salivary cortisol at awakening and in the morning. Reductions were also observed in 24-h urinary cortisol and hair cortisol. In pharmacological challenge tests, patients exhibited impaired cortisol release in response to adrenocorticotropic hormone (ACTH) stimulation and exaggerated suppression following glucocorticoid administration.
Collectively, these alterations indicate reduced free cortisol availability and enhanced HPA-axis negative feedback sensitivity, consistent with a hyporeactive endocrine state in ME/CFS. This neuroendocrine hypo-reactivity may underlie hallmark clinical features such as unrefreshing sleep, post-exertional malaise, and severe fatigue, as well as cognitive slowing, emotional blunting, and diminished stress resilience frequently observed in ME/CFS and related functional disorders.
Integrating neuroendocrine and psychological perspectives may help clarify mechanisms of chronic stress maladaptation and inform psychobiological interventions for fatigue syndromes.
Web | DOI | PDF | Molecular Psychiatry | Paywall
Woo, Tae-Wook; Choi, Yu-Jin; Kim, Jun-Yeol; Lee, Jin-Seok; Son, Chang-Gue
Abstract
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is a major clinical challenge as a complex multisystemic disorder with no well-established pathophysiological mechanism, characterized by persistent fatigue and post-exertional malaise, along with unrefreshing sleep, cognitive impairment, and impaired stress recovery.
Despite decades of investigation into the hypothalamic-pituitary-adrenal (HPA) axis, a definitive neuroendocrine hallmark has remained elusive due to inconsistent findings across various cortisol matrices. Therefore, this systematic review and meta-analysis aimed to provide an integrated understanding of HPA-axis regulation in ME/CFS.
We identified 46 case–control studies (comprising 46 independent datasets, including 12 pharmacological challenge studies), involving 1388 ME/CFS patients (71.9% female; mean age 37.3 ± 6.2 years) and 1349 matched healthy controls.
Meta-analyses showed lower salivary cortisol at awakening and in the morning. Reductions were also observed in 24-h urinary cortisol and hair cortisol. In pharmacological challenge tests, patients exhibited impaired cortisol release in response to adrenocorticotropic hormone (ACTH) stimulation and exaggerated suppression following glucocorticoid administration.
Collectively, these alterations indicate reduced free cortisol availability and enhanced HPA-axis negative feedback sensitivity, consistent with a hyporeactive endocrine state in ME/CFS. This neuroendocrine hypo-reactivity may underlie hallmark clinical features such as unrefreshing sleep, post-exertional malaise, and severe fatigue, as well as cognitive slowing, emotional blunting, and diminished stress resilience frequently observed in ME/CFS and related functional disorders.
Integrating neuroendocrine and psychological perspectives may help clarify mechanisms of chronic stress maladaptation and inform psychobiological interventions for fatigue syndromes.
Web | DOI | PDF | Molecular Psychiatry | Paywall