jnmaciuch
Senior Member (Voting Rights)
Microglial reactivity and neuroinflammation-driven changes in motivational behaviors are regulated by Orai1 calcium channels
Abstract
Microglia are the brain’s resident immune cells that respond to injury and disease by transitioning between homeostatic and reactive states. These cell state transitions determine whether microglia promote or resolve inflammation in the central nervous system (CNS). In this study, we explored the role of Ca 2+ signaling in regulating broader microglial cell state transitions and identified Orai1 Ca 2+ channels as critical regulators of microglial plasticity and neuroinflammatory signaling. Conditional deletion of Orai1 in microglia impaired their ability to adopt reactive, proinflammatory states.
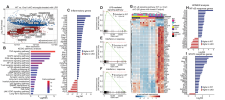
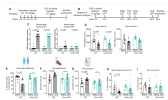
Transcriptomic and metabolomic profiling revealed that Orai1 deletion suppressed the expression of proinflammatory genes linked to immunity, inflammation, and cell metabolism. Conversely, Orai1-deficient microglia generated greater amounts of neuroprotective and anti-inflammatory mediators, including BDNF, ARG1, and the mitochondrial metabolite itaconate. In a model of CNS inflammation induced by peripheral lipopolysaccharide (LPS) challenge, microglial Orai1 deletion attenuated microglial and astrocyte reactivity and reduced hippocampal amounts of the proinflammatory cytokines IL-1β and IL-6.
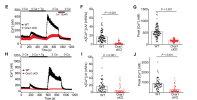
Consistent with these cellular changes, microglial Orai1 knockout mice were protected against LPS-induced decreases in motivational behaviors, including impaired reward-seeking and escape behaviors. These findings establish Orai1 channels as key regulators of microglial cell state transitions, linking Ca 2+ signaling to neuroinflammation and inflammation-driven behavioral dysfunction.
[Paragraph breaks added]
Web | DOI | Science Signaling
DeMeulenaere, Kaitlyn E.; Grant, Rogan A.; Martin, Megan E.; Valencia, Hiam A.; Radulovic, Jelena; Salter, Michael W.; Prakriya, Murali
Abstract
Microglia are the brain’s resident immune cells that respond to injury and disease by transitioning between homeostatic and reactive states. These cell state transitions determine whether microglia promote or resolve inflammation in the central nervous system (CNS). In this study, we explored the role of Ca 2+ signaling in regulating broader microglial cell state transitions and identified Orai1 Ca 2+ channels as critical regulators of microglial plasticity and neuroinflammatory signaling. Conditional deletion of Orai1 in microglia impaired their ability to adopt reactive, proinflammatory states.
Transcriptomic and metabolomic profiling revealed that Orai1 deletion suppressed the expression of proinflammatory genes linked to immunity, inflammation, and cell metabolism. Conversely, Orai1-deficient microglia generated greater amounts of neuroprotective and anti-inflammatory mediators, including BDNF, ARG1, and the mitochondrial metabolite itaconate. In a model of CNS inflammation induced by peripheral lipopolysaccharide (LPS) challenge, microglial Orai1 deletion attenuated microglial and astrocyte reactivity and reduced hippocampal amounts of the proinflammatory cytokines IL-1β and IL-6.
Consistent with these cellular changes, microglial Orai1 knockout mice were protected against LPS-induced decreases in motivational behaviors, including impaired reward-seeking and escape behaviors. These findings establish Orai1 channels as key regulators of microglial cell state transitions, linking Ca 2+ signaling to neuroinflammation and inflammation-driven behavioral dysfunction.
[Paragraph breaks added]
Web | DOI | Science Signaling