Human Endogenous Retroviruses and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Emerging Insights into Their Role in Disease Pathogenesis, Immune Dysregulation, and as Potential Biomarkers and Therapeutic Targets
Perera, Krishani Dinali; Garcia, Elisa Oltra; Carding, Simon R.
Abstract
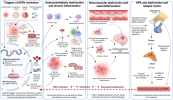
Human endogenous retroviruses (HERVs) are potential driving forces of the pathophysiology of Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), linking post-infectious immune dysfunction to chronic inflammation and immune and neurocognitive dysfunction that are hallmark features of ME/CFS.
Accumulating evidence from related autoimmune diseases and cancers has shown that reactivated HERVs can contribute to disease pathogenesis by amplifying immune activation through viral protein-mediated innate sensing, Long Terminal Repeat (LTR)-driven transcription, and disrupting epigenetic silencing. HERV signatures are therefore promising biomarkers for diagnosis, patient stratification for drug-repurposing trials, and therapy monitoring.
Accumulating evidence suggests a possible correlation between HERV expression and ME/CFS symptom severity, alterations of immune phenotypes, function and inflammatory gene networks. Importantly, locus-specific HERV profiling is a promising approach for distinguishing ME/CFS from overlapping or co-morbid conditions and healthy controls. Furthermore, HERV-targeted antibodies, immune modulators, epigenetic and antiviral interventions offer promise as concomitant therapeutic strategies for ME/CFS.
Additional research incorporating viromics and other-omics validation, functional assays, and HERV-stratified clinical trials is now needed to realise this potential and to transform ME/CFS from a symptom-based syndrome into a mechanism-driven, treatable condition.
Web | DOI | Preprints.org | Preprint
Perera, Krishani Dinali; Garcia, Elisa Oltra; Carding, Simon R.
Abstract
Human endogenous retroviruses (HERVs) are potential driving forces of the pathophysiology of Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), linking post-infectious immune dysfunction to chronic inflammation and immune and neurocognitive dysfunction that are hallmark features of ME/CFS.
Accumulating evidence from related autoimmune diseases and cancers has shown that reactivated HERVs can contribute to disease pathogenesis by amplifying immune activation through viral protein-mediated innate sensing, Long Terminal Repeat (LTR)-driven transcription, and disrupting epigenetic silencing. HERV signatures are therefore promising biomarkers for diagnosis, patient stratification for drug-repurposing trials, and therapy monitoring.
Accumulating evidence suggests a possible correlation between HERV expression and ME/CFS symptom severity, alterations of immune phenotypes, function and inflammatory gene networks. Importantly, locus-specific HERV profiling is a promising approach for distinguishing ME/CFS from overlapping or co-morbid conditions and healthy controls. Furthermore, HERV-targeted antibodies, immune modulators, epigenetic and antiviral interventions offer promise as concomitant therapeutic strategies for ME/CFS.
Additional research incorporating viromics and other-omics validation, functional assays, and HERV-stratified clinical trials is now needed to realise this potential and to transform ME/CFS from a symptom-based syndrome into a mechanism-driven, treatable condition.
Web | DOI | Preprints.org | Preprint