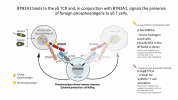
Could you explain why I get the following associations in STRING :
I don't know anything about these proteins, but if you want to see how they're related, you should be able to click the lines between proteins on the STRING plot, and it'll give you all the evidence it based the relationships on.I am particularly interested for the relations of MFGE8 (Milk Fat Globule - existing below in the figure describing efferocytosis) with BTN2A2, MERTK and GAS6.